Are NAD and FAD nucleotides
 Matthew Wilson
Matthew Wilson FAD is flavin adenine dinucleotide, and NAD is nicotinamide adenine dinucleotide. … The nucleotides in NAD are joined together by phosphate groups. Flavin adenine dinucleotide is considered a redox cofactor that is related to many metabolic reactions.
Is FAD a nucleotide?
Flavin-adenine dinucleotide (FAD) The nucleotides in nucleic acids (DNA and RNA) are also linked to each other by such bonds. FMN is a derivative of riboflavin (= vitamin B2). Thanks to the flavin group, FAD can act as a cofactor for enzymes that catalyze certain complicated redox processes, e.g. in the Krebs cycle.
What type of molecules are NAD and FAD?
NAD+ and FAD are coenzymes, organic molecules that serve as helpers during enzyme-catalyzed reactions, and they receive electrons and protons as part of these reactions.
Is NAD a nucleotide?
Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine nucleobase and the other nicotinamide. … These electron transfer reactions are the main function of NAD.Is NAD a nucleic acid?
Nicotinamide adenine dinucleotide (NAD+): A nucleic acid coenzyme used by organisms as the oxidant in certain redox reactions. … The products are a β-keto acid and NADH (the reduced form of NAD+).
Where are NAD and FAD found?
FAD is flavin adenine dinucleotide, and NAD is nicotinamide adenine dinucleotide. 2. Nicotinamide adenine dinucleotide is a coenzyme that is found in all living beings. Flavin adenine dinucleotide is considered a redox cofactor that is related to many metabolic reactions.
What is NAD and FAD?
Nicotinamide Adenine Dinucleotide (NAD) and Flavin Adenine Dinucleotide (FAD) are coenzymes involved in reversible oxidation and reduction reactions. … Then, these reduced coenzymes can donate these electrons to some other biochemical reaction normally involved in a process that is anabolic (like the synthesis of ATP).
Is FAD a cofactor or coenzyme?
FAD or flavin adenine dinucleotide is a very common coenzyme (a cofactor made up of organic molecules) in proteins. Similar to NAD and NADP in that it carries electrons, FAD participates in many important chemical reactions that flavoproteins carry out.Is FAD an oxidizing agent?
FAD has a more positive reduction potential than NAD+ and is a very strong oxidizing agent. The cell utilizes this in many energetically difficult oxidation reactions such as dehydrogenation of a C-C bond to an alkene.
Is FAD a coenzyme?FAD is an essential coenzyme for 5,10-methylene tetrahydrofolate reductase, a key enzyme of the folate activation pathway, catalyzing the interconversion of 5,10-methylene tetrahydrofolate and 5-methyltetrahydrofolate.
Article first time published onAre NAD and FAD electron acceptors?
NAD+ and FAD are oxidizing agents-that is, electron acceptors. Several different oxidation reactions of catabolism are catalyzed by en4rmes that use either NAD+ or FAD as their cofactor. 1a) fhe oxidized form of nicotinamide adenine dinucleotide. … Hence its abbreviation is NAD+.
Is NAD+ an oxidizing agent?
The cofactor is, therefore, found in two forms in cells: NAD+ is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction, also with H+, forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD.
What are the roles of NAD+ and FAD and oxygen in aerobic respiration?
In aerobic respiration, the final electron acceptor for the electron transport chain is an oxygen molecule, O2. … When NADH or FADH2 give their high energy electrons to the electron transport chain, NAD+ and FAD are regenerated.
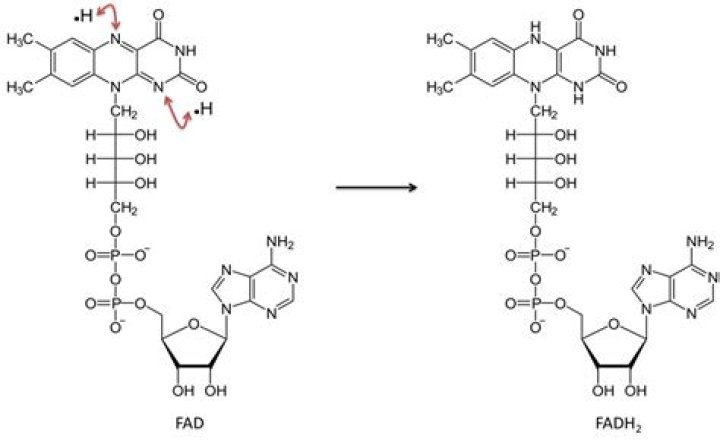
Is FAD oxidized or reduced?
Flavin adenine dinucleotide (FAD) is an important redox cofactor involved in many reactions in metabolism. The fully oxidized form, FAD, is converted to the reduced form, FADH2 by receiving two electrons and two protons. … Many proteins contain a flavin moiety, either in the form of FAD or FMN.
Is NAD+ oxidized or reduced in glycolysis?
In the process of glycolysis, NAD+ is reduced to form NADH + H+. If NAD+ is not present, glycolysis will not be able to continue. During aerobic respiration, the NADH formed in glycolysis will be oxidized to reform NAD+ for use in glycolysis again.
Is NAD+ a substrate or product?
NAD+ (nicotinamide adenine dinucleotide) is a substrate. NAD+ is utilised as a co-substrate in non-redox reactions and plays an essential role in signalling and regulatory pathways.
Are NAD and FAD oxidizing agents?
Rather they use nicotinamide adeninine dinucleotide (NAD+) or flavin adenine dinucleotide (FAD) as oxidizing agents, which get reduced. Enzymes that uses these oxidizing agents are usally called dehydrogenases. Dioxygen can also be used to introduce oxygen atoms into biological molecules in oxidative reactions.
What is a characteristic difference between FAD and NAD+ quizlet?
NAD+ transfers two electrons while FAD can transfer one or two. NAD+ transfers two electrons while FAD can transfer one or two.
What vitamins are NAD+ and FAD derived from?
Two common cofactors that are derived from the B vitamins, niacin and riboflavin, are nicotinamide adenine dinucleotide (NAD) and flavin adenine dinucleotide (FAD), respectively.
Where is FAD used in cellular respiration?
Flavin adenine dinucleotide, or FADH2, is a redox cofactor that is created during the Krebs cycle and utilized during the last part of respiration, the electron transport chain. Nicotinamide adenine dinucleotide, or NADH, is a similar compound used more actively in the electron transport chain as well.
What is the function of molecules like NAD +/ NADH and FAD FADH2 in cells?
Both NAD+/NADH and FAD/FADH2 are extensively used in energy extraction from sugars during catabolism in chemoheterotrophs, whereas NADP+/NADPH plays an important role in anabolic reactions and photosynthesis.
Is NAD+ organic or inorganic?
They are divided into organic molecules = coenzymes, and inorganic elements = metal ions. Coenzymes= NAD+ (nicotinamide adenine dinucleotide), FAD (flavin adenine dinucleotide), CoA (coenzyme A) Metal ions = Iron, copper, calcium, zinc, magnesium.
Is FAD charged?
The reduced coenzyme FADH2 contributes to oxidative phosphorylation in the mitochondria. FADH2 and NADH can be considered to be like “charged batteries” from having accepted electrons and a proton or two. FADH2 is reoxidized to FAD, which makes it possible to produce two molecules of the universal energy carrier ATP.
Is NAD a cofactor or coenzyme?
Nicotinamide adenine dinucleotide (NAD) is one of the most important coenzymes in the cell. Not surprisingly, NAD and the closely related NADP are the two most abundant cofactors in eukaryotic cell. Healthy bodies make all the NADH they need using vitamin B3 (also known as niacin, or nicotinamide) as a starting point.
What role does NAD and FAD play in cellular respiration?
Both NAD and FAD play a crucial role in cellular respiration to temporarily store energy as it’s released from glucose. Transferring the electrons NAD and FAD are carrying during an oxidation reaction releases the stored energy that was harvested from glucose.
What type of cofactor is FAD?
4 FAD. FAD is a redox cofactor of several important reactions in metabolism. This cofactor exists in two different redox states, with FAD and FADH2 being the oxidized and reduced forms, respectively. FAD is formed of a riboflavin moiety (vitamin B2), coupled to a phosphate group of an ADP molecule.
What are the examples of fads?
A fad is a product that has a very brief product life cycle that rises quickly in popularity but then declines about as quickly. Examples of fads include the Hula-hoop®, Pet Rock®, Pokémon®, the yo-yo and Beanie Babies®.
Which metabolic pathway requires both NAD+ and FAD?
Cellular respiration involves a metabolic pathway of enzymes assisted by coenzymes . The two coenzymes involved in cellular respiration, NAD+ and FAD, receive the hydrogen atoms removed from glucose. Glucose has 12 hydrogen atoms that will be pulled off one at a time and picked up by NAD+ or FAD.
Why is FAD used instead of NAD+?
Succinate dehydrogenase oxidizes succinate to produce fumarate. FAD is used as the hydrogen acceptor, instead of NAD+. The free-energy change of the reaction is insufficient to reduce NAD+.
What is NAD in respiration?
The cellular respiration processes of all living cells make use of the coenzyme Nicotinamide adenine dinucleotide (NAD). It plays a key role in energy metabolism by accepting and donating electrons. … NADH contributes to oxidation in cell processes like glycolysis to help with the oxidation of glucose.
Is NAD+ The oxidized form?
Generally, NAD exists in two forms such as reduced and oxidized forms. The reduced form of NAD is designated as NADH and oxidized form as NAD+. Each form helps to carry electrons from one reaction to another. It also plays a vital role in energy production via redox reactions.





