What are the 5 Strong acids
 Natalie Ross
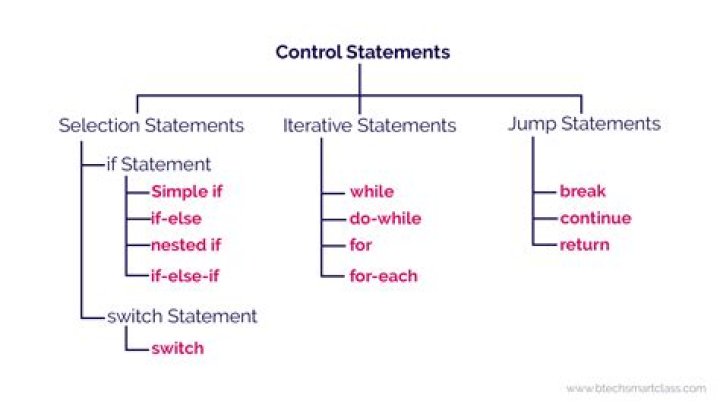
Natalie Ross HCl (hydrochloric acid)HNO3 (nitric acid)H2SO4 (sulfuric acid)HBr (hydrobromic acid)HI (hydroiodic acid)HClO3 (chloric acid)HClO4 (perchloric acid)
What are 7 strong acids?
- Dissociation of Strong Acids and Weak Acids.
- Chloric Acid (HClO3)
- Hydrobromic Acid (HBr)
- Hydrochloric Acid (HCl)
- Hydroiodic Acid (HI)
- Nitric Acid (HNO3)
- Perchloric Acid (HClO4)
- Sulfuric Acid (H2SO4)
What are 5 weak acids?
- Formic acid (chemical formula: HCOOH)
- Acetic acid (chemical formula: CH3COOH)
- Benzoic acid (chemical formula: C6H5COOH)
- Oxalic acid (chemical formula: C2H2O4)
- Hydrofluoric acid (chemical formula: HF)
- Nitrous acid (chemical formula: HNO2)
- Sulfurous acid (chemical formula: H2SO3)
What are the 5 acids?
The five common acids have several uses in medicine, industries, and at home. The five common acids are sulfuric acid, hydrochloric acid, nitric acid, lactic acid, and acetic acid.What are the strong acids in order?
There are 7 strong acids: chloric acid, hydrobromic acid, hydrochloric acid, hydroiodic acid, nitric acid, perchloric acid, and sulfuric acid.
What are the 6 strong acids and bases?
- HCl – hydrochloric acid.
- HNO3 – nitric acid.
- H2SO4 – sulfuric acid.
- HBr – hydrobromic acid.
- HI – hydroiodic acid.
- HClO4 – perchloric acid.
What are the 6 strong acids?
For the MCAT, you should know that strong acids are acids that dissociate completely in solution. There are six of them that you have to have memorized for the MCAT. They are H2SO4 (or sulfuric acid), HI (hydrologic acid), HBr (hydrobromic acid), HNO3 (nitric acid), HCl (hydrochloric acid) and HClO4 (perchloric acid).
What are 5 acids and 5 bases?
- Acetic Acid (HC₂H₃O₂)
- Citric Acid (H₃C₆H₅O₇)
- Lactic Acid (C₃H₆O₃)
- Sulfuric Acid (H₂SO₄)
- Ascorbic Acid (C₆H₈O₆)
What are the top 10 acids?
- Recommended Videos. …
- Hydrochloric Acid (HCl) …
- Hydrobromic Acid (HBr) …
- Hydroiodic Acid (HI) …
- Sulphuric Acid (H2SO4) …
- Nitric Acid (HNO3) …
- Perchloric Acid (HClO4) …
- Chloric Acid (HClO3) Chloric acid is a chemical compound with the formula HClO3.
- of 11. Acetic Acid. Acetic acid is also known as ethanoic acid. …
- of 11. Boric Acid. This is the chemical structure of boric acid: boron (pink), hydrogen (white) and oxygen (red). …
- of 11. Carbonic Acid. …
- of 11. Citric Acid. …
- of 11. Hydrochloric Acid. …
- of 11. Hydrofluoric Acid. …
- of 11. Nitric Acid. …
- of 11. Oxalic Acid.
What is an example of a strong acid?
Examples of Strong Acids Hydrobromic acid (HBr): pKa = -8.7. Perchloric acid (HClO4): pKa ≈ -8. Hydrochloric acid (HCl): pKa = -6.3. Sulfuric acid (H2SO4): pKa1 ≈ -3 (first dissociation only)
What are some examples of strong acids?
Strong AcidsStrong Baseshydrobromic acid (HBr)potassium hydroxide (KOH)hydroiodic acid (Hl)calcium hydroxide (Ca(OH)2)nitric acid (HNO3)strontium hydroxide (Sr(OH)2)sulfuric acid (H2SO4)barium hydroxide (Ba(OH)2)
Is Vinegar a strong acid?
Acetic acid makes vinegar mildly acidic, with a typical pH of 2–3.
What are strong acids Class 10?
An acid which dissociates completely in water are strong acids and an acid which dissociates almost completely in water are weak acids.
What are the 8 strong bases?
- LiOH (lithium hydroxide)
- NaOH (sodium hydroxide)
- KOH (potassium hydroxide)
- Ca(OH)2 (calcium hydroxide)
- RbOH (rubidium hydroxide)
- Sr(OH)2 (strontium hydroxide)
- CsOH (cesium hydroxide)
- Ba(OH)2 (barium hydroxide)
What are 10 common household acids?
- Vinegar – weak acetic acid.
- Lemon juice – citric acid and some ascorbic acid.
- Any citrus fruit – citric acid and some ascorbic acid.
- Most other fruits – citric acid, possibly tartaric, oxalic, or malic acid.
- Carbonated soda – phosphoric, carbonic, and sometimes citric acid.
- Battery acid – sulfuric acid.
Is NH3 a base?
Ammonia, NH3, is a Lewis base and has a lone pair. It will donate electrons to compounds that will accept them. Donation of ammonia to an electron acceptor, or Lewis acid. There may be anionic or neutral Lewis bases.
Is HClO3 a strong acid?
The 7 common strong acids are: HCl, HBr, HI, HNO3, HClO3, HClO4 and H2SO4 (1st proton only). … They simply do not ionize completely whereas a strong acid or base does.
How many strong bases are there?
List of Strong Bases. Like strong acids, strong bases also dissolve thoroughly in water, as opposed to weak bases that do not dissolve. Strong bases release hydroxide (OH-) ions and absorb hydrogen (H+) molecules. There are eight strong bases.
Which is highly strong acid?
Strong AcidsStrong Baseshydrobromic acid (HBr)potassium hydroxide (KOH)hydroiodic acid (Hl)calcium hydroxide (Ca(OH)2)nitric acid (HNO3)strontium hydroxide (Sr(OH)2)sulfuric acid (H2SO4)barium hydroxide (Ba(OH)2)
Which is most strong acid?
The world’s strongest superacid is fluoroantimonic acid. Fluoroantimonic acid is a mixture of hydrofluoric acid and antimony pentafluoride. The carbonane superacids are the strongest solo acids.
What are the 3 types of acids?
Usually acids can be divided into three major types. First one is binary acid, second one is oxyacid, and the last one is carboxylic acid. Binary acids are all written in “H-A” form, which means hydrogen bond to a nonmetal atom.
What are 5 examples of acids and bases?
ACIDSBASESproduce carbon dioxide when reacted with carbonates.Common examples: Lemons, oranges, vinegar, urine, sulfuric acid, hydrochloric acidCommon Examples: Soap, toothpaste, bleach, cleaning agents, limewater, ammonia water, sodium hydroxide.
What are 5 examples of bases?
- Potassium hydroxide (KOH)
- Sodium hydroxide (NaOH)
- Barium hydroxide (Ba(OH)2)
- Caesium hydroxide (CsOH)
- Strontium hydroxide (Sr(OH)2)
- Calcium hydroxide (Ca(OH)2)
- Lithium hydroxide (LiOH)
- Rubidium hydroxide (RbOH)
What are 5 properties of bases?
- Bases change the colour of litmus from red to blue.
- They are bitter in taste.
- Bases lose their basicity when mixed with acids.
- Bases react with acids to form salt and water. …
- They can conduct electricity.
- Bases feel slippery or soapy.
- Some bases are great conductors of electricity.
What are called strong acids?
Examples of Strong Acids HCl (hydrochloric acid) … HNO3 (nitric acid) HBr (hydrobromic acid) HClO4 (perchloric acid) HI (hydroiodic acid)
What are concentrated acids?
A concentrated acid is one in which many acid molecules are dissolved in a set volume of solution, while a dilute acid will have very few molecules per unit volume. … By adding water to a concentrated acid or base, the solution is diluted. The more water that is added the more dilute the solution becomes.
Which list contains only strong acids?
The strong acids are hydrochloric acid, nitric acid, sulfuric acid, hydrobromic acid, hydroiodic acid, perchloric acid, and chloric acid. The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid (HF).
Why is HCl a strong acid?
Dissociation of acids When HCl molecules dissolve they dissociate into H+ ions and Cl- ions. HCl is a strong acid because it dissociates almost completely. By contrast, a weak acid like acetic acid (CH3COOH) does not dissociate well in water – many H+ ions remain bound-up within the molecule.
What acid is in milk?
The real acidity of milk is due to lactic acid.
Is soap an acid?
Hint :Soap is made up of a weak acid (fatty acids) and a strong base (lye), resulting in what is known as “alkali salt,” or a salt with a basic pH. When you put a pH strip (also known as a litmus test) in soapy water, it usually comes back with an 8 or 9 .