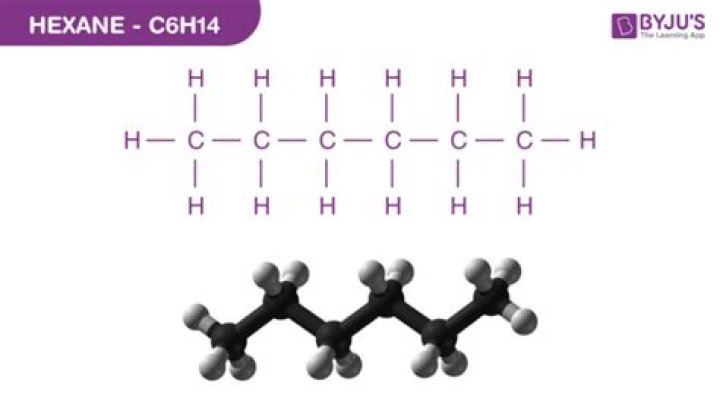
What are the uses of hexane
 James Williams
James Williams Extracting edible oils from seeds and vegetables.As an additive in consumer products including gasoline, glue, varnishes and inks.As a cleaning agent in the textile, furniture and printing industries;As a special glue used in roofing, shoemaking and leather products.
What is hexane used for?
Hexane is used to extract edible oils from seeds and vegetables, as a special-use solvent, and as a cleaning agent. Acute (short-term) inhalation exposure of humans to high levels of hexane causes mild central nervous system (CNS) effects, including dizziness, giddiness, slight nausea, and headache.
Is hexane a poison?
In humans, n-hexane is of low acute toxicity. No cases of lethality were reported after inhalation of n-hexane or commercial hexane.
What is special about hexane?
Hexane is a solvent widely used as an industrial cleaner and degreaser and is an ingredient in many consumer products. Easily inhaled or absorbed through the skin, hexane has been recognized for more than 40 years to cause long-lasting and even permanent nerve damage in feet, legs, hands, and arms.Is hexane used as a fuel?
n-Hexane is a highly volatile component of the paraffin (also the alkane or aliphatic) fraction of crude oil and natural gas, and it is a constituent of heating and motor fuels refined from petroleum.
Why we use hexane as a solvent?
So why is hexane used for extraction? Because hexane evaporates quickly and will boil at 154.4 degrees Fahrenheit, in addition to having low toxicity levels, hexane is the preferred solvent for both methods of extraction.
Why is hexane used for oil?
Hexane has been widely used for oil extraction because of easy oil recovery, narrow boiling point (63–69 °C) and excellent solubilizing ability [3]. In contrary, while in extraction and recovery processes, hexane is released into the environment that react with the pollutants to form ozone and photo chemicals [4].
Is hexane an organic solvent?
Overview. Organic solvents are carbon-based substances capable of dissolving or dispersing one or more other substances. … Organic solvents recognized as neurotoxins include n-hexane, tetrachloroethylene, and toluene.What products contain hexane?
It is used as a cleaning agent in printing, shoe making, textiles, automotive brake repair, and furniture making. It is also used in the food industry. Common household products, such as spray adhesives, contact cement, arts and craft paints, and stain removers contain hexane.
Is hexane cancerous?There is no evidence that n-hexane causes cancer in people or animals. The Department of Health and Human Services (DHHS), International Agency for Research on Cancer (IARC) and the EPA have not classified n-hexane for carcinogenicity.
Article first time published onWhy is hexane toxic?
Enzymes in your liver break down n-hexane. If you are exposed to high concentrations of n-hexane over a long period, one of these breakdown products may cause damage to your nervous system. Most of these breakdown products leave your body in the urine within a day or two.
Does hexane dissolve plastic?
n-Hexane can react vigorously with oxidizing materials such as liquid chlorine, concentrated oxygen, and sodium hypochlorite. n-Hexane will attack some forms of plastics, rubber, and coatings.
Why hexane is used in gas chromatography?
It is as simple as the compounds that you want to separate and identify being soluble in the solvent. Generally low boiling solvents, such as hexane or methanol, are used as they move rapidly through the column. … Generally, a solvent is chosen with a much lower boiling than the compounds.
Why can oil dissolve in hexane?
Because oil is a strongly non-polar compound, solvents that dissolve it are highly non-polar, and may feature an organic formulation that is high in volatile organic compounds (VOCs). … Hexane — Frequently used as a solvent for dissolving various types of cooking oil.
Is hexane used to extract olive oil?
Olive oil extraction is the process of extracting the oil present in olive drupes, known as olive oil. … This contrasts with other oils that are extracted with chemical solvents, generally hexane.
Is hexane used to make olive oil?
Hexane extraction is a form of oil extraction that uses the chemical solvent hexane and evaporation to pull the oils from various oil seeds, fruits, nuts, and olives to create cooking oils. … This is the fastest and easiest method to extract a large amount of oil in a short amount of time.
Is hexane in vegetable oil?
As with many highly processed food products there are concerns about the safety of canola oil. It has been estimated that refined vegetable oils extracted with hexane contain approximately 0.8 milligrams of residual hexane per kilogram of oil (0.8 ppm). … Deodorization is the final step in refining ALL vegetable oils.
Is hexane used to extract peanut oil?
During solvent extraction, hexane is used to wash the flakes. After extraction, the hexane in the extracted oil and the hexane in remaining flakes is evaporated, condensed, and recovered for reuse. In this experiment we use hexane to extract peanut oil; however we will not recover used hexane.
Is hexane used in refined oil?
Hexane is one of the most commonly used solvents in the edible oil industry 1. … Since refining, bleaching and deodorization are carried out as complementary processes before using oil as food products, it is likely that solvent residue approximately eliminated 7.
Is hexane a better solvent than water?
Type of SubstanceSoluble in Water?Soluble in Hexane?Molecular Compounds with Nonpolar MoleculesNoYes
What is hexane in food?
Hexane is a solvent made from crude oil. In the food industry, hexane is used to extract the vegetable oil from plant seeds such as canola, soybeans, sunflowers, and corn because it is more efficient and less expensive than squeezing out the oil with presses.
Is hexane bad for hair?
Hexane is not actually used as an ingredient, but as a solvent to extract natural oils used in haircare and skincare products. When contaminated with hexane, your beauty products can be harmful in the long run. Aside from inhalation, hexane can also affect you by passing through the skin.
Does hexane remove paint?
Consumer Applications. Consumer products containing Hexane include rubber cement, quick drying glues, gasoline, and paint remover.
Is hexane an acid or base?
You may call it “effective pH” or like that, but remember: it has little to do with literal concentration of H+, and your hypothetical solution of 10−30M HCl in hexane is in fact neither acidic nor basic.
Where is hexane found?
What is Hexane and Where Can it Be Found? Hexane is a chemical commonly extracted from petroleum and crude oil. It is a colorless liquid that gives off a subtle, gasoline-like odor. Hexane is highly flammable, yet it can be found in many household products such as stain removers for arts and crafts projects.
Why is hexane a liquid at room temperature?
In HEXANE, because London forces increase with molecular mass, at room temperature dispersion forces > kinetic energy, so hexane is a liquid. However, as temperature is raised, kinetic energy increases and at a certain point hexane will become a gas.
Can hexane cause infertility?
It is not known if exposure to n-hexane can affect fertility in people. Experiments done with animals that were fed or breathed in n-hexane did not show any effect on fertility. There is no evidence that exposure to n-hexane increases the risk of cancer in people.
What is hexane free?
Hexane Free Castor Oil is light yellow translucent oil that originated from the castor beans. The cold-pressed technique used for the extraction of castor oil, is free from harmful impurities. Castor oil is rich in vitamins, fatty acids, and Ricinoleic acid, which have a lot of benefits for health and beauty.
What makes hexane flammable?
Hexane is flammable because the two constituent elements, carbon, and hydrogen, are both much happier to react with oxygen (to form water and carbon dioxide) than they are with each other. This is due to the electrochemical makeup of the elements themselves and oxygen’s strong ability to “oxidize” other elements.
Is hexane more flammable than gasoline?
Fuel Gas”Lower Explosive or Flammable Limit” (LEL/LFL) (% by volume of air)”Upper Explosive or Flammable Limit” (UEL/UFL) (% by volume of air)Hexane1.17.5
Does hexane flammable?
n-Hexane is a FLAMMABLE LIQUID. DO NOT attempt to extinguish fire unless flow can be stopped.





