What does a large kcat mean
 Leah Mitchell
Leah Mitchell One way to measure the catalytic efficiency of a given enzyme is to determine the kcat/km ratio. … The greater the ratio, the higher the rate of catalysis is; conversely, the lower the ratio, the slower the catalysis is.
What does a high kcat km mean?
One way to measure the catalytic efficiency of a given enzyme is to determine the kcat/km ratio. … The greater the ratio, the higher the rate of catalysis is; conversely, the lower the ratio, the slower the catalysis is.
What does it mean when kcat increases?
It is (kr+kcat)/kf. To compensate for the higher Km, kcat or the turnover number or conversion of the Enzyme substrate complex to enzyme plus product must increase if the kcat/Km is the same. So in essence what you say is correct as either kf must go down, or kr must go up to make a higher Km, if kcat is higher.
Is a higher kcat better?
If the enzyme has more than one possible substrate, the kcat/Km values determine the specificity of the enzyme for each. The higher this value the more specific the enzyme is for that substrate. This is because a high value of kcat and a low value of Km are expected for the best substrates.What is the significance of kcat?
Kcat is the turnover number — the number of substrate molecule each enzyme site converts to product per unit time. If you know the concentration of enzyme sites, you can fit Kcat instead of Vmax when analyzing a substrate vs. velocity curve.
What is Ki Biochem?
Ki, the inhibitor constant The inhibitor constant, Ki, is an indication of how potent an inhibitor is; it is the concentration required to produce half maximum inhibition. Plotting 1/v against concentration of inhibitor at each concentration of substrate (the Dixon plot) gives a family of intersecting lines.
What does higher km mean?
This is usually expressed as the Km (Michaelis constant) of the enzyme, an inverse measure of affinity. … An enzyme with a high Km has a low affinity for its substrate, and requires a greater concentration of substrate to achieve Vmax.”
What is kcat MCAT?
kcat is also known as the turnover number of the enzyme – defining the maximum number of substrate molecules. converted to product per unit of time. kcat = Vmax/[ET]Is kcat the same as K2?
Kcat is equal to K2, and it measures the number of substrate molecules “turned over” by enzyme per second. … The higher the Kcat is, the more substrates get turned over in one second. Km is the concentration of substrates when the reaction reaches half of Vmax.
Is a small kcat better?The smaller the dissociation constant is, the better two reactants can combine. Since the affinity of enzyme with substrate determines how favorable the reaction can form enzyme-substrate complex, kd is often studied in Michaelis-Menten equation.
Article first time published onWhat can affect kcat?
As Km is constant, the affinity of the enzyme for the substrate should not change. therefore what has changed probably is the structure of the active site. And this change of structure causes Kcat to increase.
Does inhibitor change kcat?
The binding site of the inhibitor is not at the active site. However, the inhibitor binding causes a change in the conformation of the protein that affects either substrate binding (Km), the chemical step (Kcat), or both.
Is kcat dependent on pH?
Soc. 81, 5907-5914] that only EH and EHS interconvert directly, the pH-dependence of kcat. is determined only by A. … The pH-dependence of Km is determined in general by B-C/A-D, but when reactants and intermediate are in equilibrium, C identical to D and this expression simplifies to B/A.
How do you find kcat?
It’s true that to calculate Kcat of an enzyme , you can use Kcat=Vmax/[Et]. However, to calculate [Et]=Total enzyme conc, you need the amount of your protein and the total volume of the enzymatic reaction.
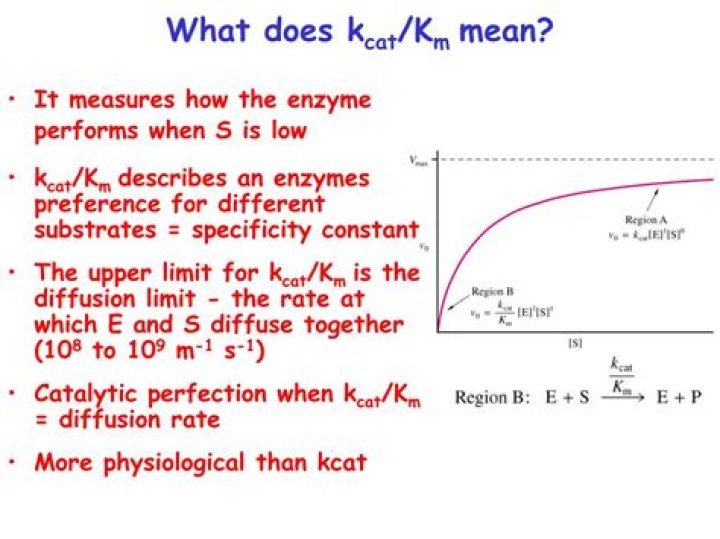
What does a high specificity constant mean?
), is a measure of how efficiently an enzyme converts substrates into products. A comparison of specificity constants can also be used as a measure of the preference of an enzyme for different substrates (i.e., substrate specificity). The higher the specificity constant, the more the enzyme “prefers” that substrate.
What does catalytic efficiency tell you?
Increasing the reaction rate of a chemical reaction allows the reaction to become more efficient, and hence more products are generated at a faster rate. … This is known as the catalytic efficiency of enzymes, which, by increasing the rates, results in a more efficient chemical reaction within a biological system.
What is the Lineweaver Burk equation?
The Lineweaver-Burk equation is a linear equation, where 1/V is a linear function of 1/[S] instead of V being a rational function of [S]. The Lineweaver-Burk equation can be readily represented graphically to determine the values of Km and Vmax. … Given a Lineweaver-Burk plot, determine the Km of a particular enzyme.
What is KMAX and Vmax?
Vmax is the maximum rate of an enzyme catalysed reaction i.e. when the enzyme is saturated by the substrate. Km is measure of how easily the enzyme can be saturated by the substrate. Km and Vmax are constant for a given temperature and pH and are used to characterise enzymes.
What is the unit of k cat?
The units of kcat are moles of product/sec divided by moles of enzyme. In consequence, kcat resulted in 1/time units. Enzyme concentration can be M, mM, uM, etc. The result of kcat*[E] is rate units: mM/min, uM/s, etc.
What is IC50 and Ki?
By definition, IC50 is the “total” concentration of inhibitor needed to reach 50% inhibition; while Ki is the “free” concentration of inhibitor required to reach 50% enzyme saturation. … Therefore, IC50 is dependent on the enzyme concentration, and is always larger than Ki.
What is pKI pharmacology?
pKI : Negative logarithm of the KI, the equilibrium dissociation constant of an antagonist-receptor complex measured in a biochemical binding or enzyme study. pD2 : Historical term for the negative logarithm of the EC50 for an agonist in a functional assay, not often used in present-day pharmacology.
Are km and KS the same?
It is extremely important to note that Km in the general equation does not equal the Ks, the dissociation constant used in the rapid equilibrium assumption! Km and Ks have the same units of molarity, however.
What is Vmax Biochem?
Vmax is the reaction rate when the enzyme is fully saturated by substrate, indicating that all the binding sites are being constantly reoccupied.
How do I find my ki?
Thus, KI can be determined by plotting the slope values vs. [I]. The resulting secondary plot or “replot” will have a Y-axis intercept of KM/Vmax and a slope of KM/VmaxKI. The value of KI is the slope/intercept of this replot.
What is k1 in Michaelis Menten?
k1 is the forward rate constant for substrate binding. k-1 is the reverse rate constant for substrate binding.
What is kcat Reddit?
Kcat = turnover of substrate to product / unit time. Kcat/Km = measure of catalytic efficiency. You use this if the question is asking about which enzyme has the greatest efficiency.
Is kcat rate limiting?
The kcat of an enzyme reflects that rate-limiting step. … However, for glycoside hydrolases the kcat/Km is an approximation of the firs step (glycosylation), that you can then relate to Ea of the first step.
What is the difference between Vmax and kcat?
Vmax & Kcat To determine Kcat, one must obviously know the Vmax at a particular concentration of enzyme, but the beauty of the term is that it is a measure of velocity independent of enzyme concentration, thanks to the term in the denominator. Kcat is thus a constant for an enzyme under given conditions.
What does kcat km mean biochemistry?
It is a measure of how many bound substrate molecules turnover or form product in 1 second. … The constant kcat/Km is also referred to as the specificity constant in that it describes how well an enzyme can differentiate between two different competing substrates.
Does kcat affect Vmax?
When you change enzyme concentration how does that affect Vmax, KM, and kcat? … Vmax depends on the enzyme concentration, so if you double the amount of enzyme you double Vmax. Km and kcat are constants so changing the enzyme concentration will not change their value.
How do you increase kcat?
To improve the kcat of an enzyme is crucial factor either you are using directed evolution, site directed mutagenesis, DNA shuffling or any method.





