What does Boyles law state
 Ava Mcdaniel
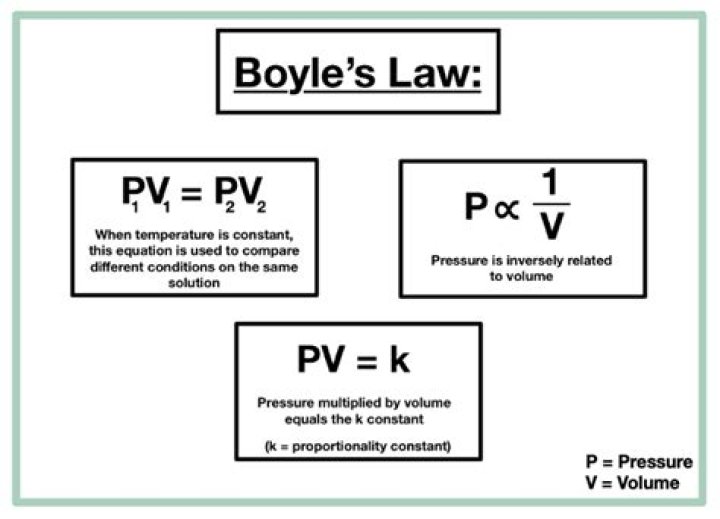
Ava Mcdaniel This empirical relation, formulated by the physicist Robert Boyle in 1662, states that the pressure (p) of a given quantity of gas varies inversely with its volume (v) at constant temperature; i.e., in equation form, pv = k, a constant. …
What is Boyles Law in simple terms?
Boyle’s Law is a basic law in chemistry describing the behavior of a gas held at a constant temperature. The law, discovered by Robert A. Boyle in 1662, states that at a fixed temperature, the volume of gas is inversely proportional to the pressure exerted by the gas.
How do we use Boyle's Law in everyday life?
If you decrease its pressure, its volume increases. You can observe a real-life application of Boyle’s Law when you fill your bike tires with air. When you pump air into a tire, the gas molecules inside the tire get compressed and packed closer together.
What does Boyle's law state for kids?
What does Boyle’s Law state? According to this law : If a fixed amount of ideal gas is kept at a fixed temperature, the pressure (P) and volume (V) are inversely proportional, that is, when one doubles, the other is reduced by half.Why is Boyle's law important?
Why is Boyle law important? Boyle’s law is significant because it explains how gases behave. It proves beyond a shadow of a doubt that gas pressure and volume are inversely proportional. When you apply pressure on a gas, the volume shrinks and the pressure rises.
What have you learned about Boyle's Law?
Robert Boyle, a famous English chemist, discovered in 1662 that if you pushed on a gas, its volume would decrease proportionately. For example, if you doubled the pressure on a gas (increase the pressure two times), its volume would decrease by half (decrease the volume two times).
How did Boyle impact society?
Robert Boyle put chemistry on a firm scientific footing, transforming it from a field bogged down in alchemy and mysticism into one based on measurement. He defined elements, compounds, and mixtures, and he coined the new term ‘chemical analysis,’ a field in which he made several powerful contributions.
What did Boyle believe about atoms?
Robert Boyle believed that all substances were made up of atoms, which could in turn exist alongside a void, or vacuum.What is the aim of Boyle's law experiment?
The primary objective of this experiment is to determine the relationship between the pressure and volume of a confined gas. The gas we will use is air, and it will be confined in a syringe connected to a Gas Pressure Sensor (see Figure 1).
What was Boyle famous quote?Robert Boyle Quotes. God would not have made the universe as it is unless He intended us to understand it. … even when we find not what we seek, we find something as well worth seeking as what we missed. From a knowledge of His work, we shall know Him.
Article first time published onHow did Boyle's research transform chemistry into real science?
How did boyle transform the science of chemistry ? Boyle established that all matter is composed of tiny particles that behave in certain know- able ways. How did newton use observations of nature to explain the movements of the planets ? … He used Plato’s emphasis on mathematics and reality to discover nature’s laws.
What is the conclusion of Boyle's law experiment?
Conclusion. If the volume of the gas decreases, the pressure of the gas increases. If the volume of the gas increases, the pressure decreases. These results support Boyle’s law.
What is the hypothesis of Boyle's Law?
The hypothesis for experiment 1, which is Boyle’s Law, is as pressure on a gas increases, the volume of the gas decreases and vice versa (V 1/P). This is an inversely proportional relationship.
How did Boyle redefine an element?
Robert Boyle. Boyle’s definition of an element was based on the observation that many substances can be decomposed into simpler substances. … They are therefore the elementary, or simplest, chemical substances. Thus, as Boyle pointed out, an element is any substance that cannot be decomposd into a simpler substance.
When did Boyle discover Boyle's Law?
One of their findings, published in 1662, later became known as “Boyle’s law.” This law expresses the inverse relationship that exists between the pressure and volume of a gas, and it was determined by measuring the volume occupied by a constant quantity of air when compressed by differing weights of mercury.
What was the significance of the works of Boyle and Lavoisier in the development of the atomic theory?
A later breakthrough in the discovery of the atomic model came through the work of French chemist Antoine Lavoisier who through a series of experiments found that the total mass of products and reactants in a chemical reactions is always the same. This led to the theory of the law of conservation of mass.
What did Boyle invent?
Robert Boyle was a 17th century chemist, philosopher, and theological writer famous for his invention of Boyle’s Law and his vacuum pump. Boyle rejected the Aristotelian emphasis on logic and theory in favor of experimental research and empirical evidence.
Where was Boyle's law discovered?
But the new generation brought the growth of experimental science in England, and the studies on air pressure culminated with the publication of BOYLE’S Law in t662.
What were Boyle's contributions to modern chemistry quizlet?
What were Boyle’s contribution to modern chemistry? Boyles stated that if a substance could be broken down into simpler substances, it was not an element.
What are the constant values needed to demonstrate Boyle's Law?
According to Boyle’s Law, an inverse relationship exists between pressure and volume. Boyle’s Law holds true only if the number of molecules (n) and the temperature (T) are both constant.
What is experimental verification of Boyle's Law?
Boyle’s law says that the pressure of a gas is inversely proportional to its volume. Below is a table of data presented by Robert Boyle in 1662. Boyle and an assistant carried out an experiment in which a parcel of gas was subjected to a steadily increased applied pressure.
What is the controlled variable in Boyle's law experiment?
These variables are P (pressure), V (volume), T (temperature), and n (number of moles). In Boyle’s experiment, which two vari- ables were held constant? Both the temperature (T) and the number of moles of gas (n) were held constant in Boyle’s J-tube experiment.
What is the dependent variable in Boyle's law experiment?
The constant, k, is dependent on temperature. The volume of an enclosed gas is inversely proportional to the pressure of the gas provided the temperature of the gas is constant.
What is Charles Law and Boyle's law?
Boyle’s law—named for Robert Boyle—states that, at constant temperature, the pressure P of a gas varies inversely with its volume V, or PV = k, where k is a constant. … Charles (1746–1823)—states that, at constant pressure, the volume V of a gas is directly proportional to its absolute (Kelvin) temperature T, or V/T = k.
Who discovered oxygen?
When Joseph Priestley discovered oxygen in 1774, he answered age-old questions of why and how things burn.