What clinical studies means
 Matthew Wilson
Matthew Wilson A type of research study that tests how well new medical approaches work in people. These studies test new methods of screening, prevention, diagnosis, or treatment of a disease. Also called clinical trial.
How does a clinical study work?
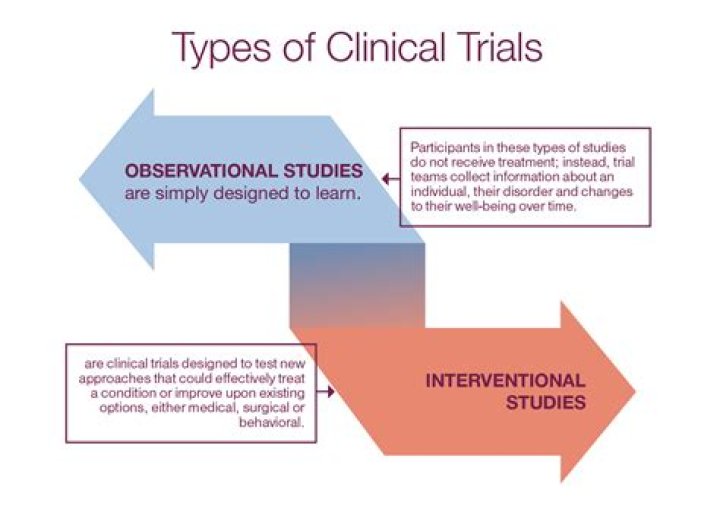
One group receives an existing treatment for a condition, and the second group receives a new treatment. Researchers then compare which group has better results. One group receives a new treatment, and the second group receives a placebo, an inactive product that looks like the test product.
What are the 5 different types of clinical trials?
There are several types of cancer clinical trials, including treatment trials, prevention trials, screening trials, supportive and palliative care trials, and natural history studies.
What are the 4 types of clinical trials?
- Pilot studies and feasibility studies.
- Prevention trials.
- Screening trials.
- Treatment trials.
- Multi-arm multi-stage (MAMS) trials.
- Cohort studies.
- Case control studies.
- Cross sectional studies.
Is clinical research a good career?
Clinical research is an attractive industry for researchers in India because it foresees enormous growth and job opportunities not only for trained medical, pharmaceutical, and paramedical professionals but also for project management staff, regulatory authorities, government, and the society at large.
How many phases of clinical studies are there?
There are 3 main phases of clinical trials – phases 1 to 3. Phase 1 trials are the earliest phase trials and phase 3 are later phase trials. Some trials have an earlier stage called phase 0, and there are some phase 4 trials done after a drug has been licensed. Some trials are randomised.
Do you get paid for clinical trials?
Clinical trials generally pay between $50-$300 per day/visit, with compensation dependant upon the length of the time required as well as the procedures performed. Overnight stays typically pay more money than those involving repeat visits.
How long is a clinical trial?
Clinical trials alone take six to seven years on average to complete. Before a potential treatment reaches the clinical trial stage, scientists research ideas in what is called the discovery phase. This step can take from three to six years.What degree do you need for clinical research?
To secure a job as a CRC, professionals must have at least a four-year bachelor’s degree in a field such as medical technology, microbiology, or public health administration. However, some employers require an additional two-year master’s degree, particularly for management positions.
Who pays for clinical trials?The sponsor of the study (such as the government, drug makers or technology companies) typically pays for all costs involved with a clinical research study. This includes supplying the new treatment, as well as any special testing, possible extra physician visits, and research costs involved in the clinical studies.
Article first time published onWhat is the most common type of clinical trial?
RankTherapy AreaTotal Number of Clinical Trials*1.Cancers361,6282.Cardiovascular & Circulatory Diseases62,1153.Infectious Diseases34,2134.Digestive Diseases31,591
What does clinical research constitute Of?
Clinical research is a branch of healthcare science that determines the safety and effectiveness (efficacy) of medications, devices, diagnostic products and treatment regimens intended for human use. These may be used for prevention, treatment, diagnosis or for relieving symptoms of a disease.
How much money does a clinical researcher make?
Annual SalaryMonthly PayTop Earners$124,500$10,37575th Percentile$100,500$8,375Average$77,425$6,45225th Percentile$54,000$4,500
How hard is clinical research?
Not only is clinical research a fulfilling career, but it’s also a challenging one. Clinical research is responsible for ascertaining how safe and effective drugs, devices and diagnostic products are, and as such it’s a demanding role with plenty of responsibility riding on it.
How much do medical researchers get paid?
The median annual wage for medical scientists was $91,510 in May 2020. The median wage is the wage at which half the workers in an occupation earned more than that amount and half earned less. The lowest 10 percent earned less than $50,240, and the highest 10 percent earned more than $164,650.
Do studies do money?
- Survey Junkie. Survey Junkie is a survey site that’s been around since 2005. …
- American Consumer Opinion. …
- Respondent. …
- FocusGroup.com. …
- Plaza Research. …
- Mindswarms. …
- Fieldwork. …
- User Interviews.
What is a Phase 5 clinical trial?
Phase 5 Clinical Trial means a post-registration clinical trial that is not required as a condition to, or for the maintenance of, any Marketing Approval or Pricing and/or Reimbursement Approval for a Licensed Product. Phase 5 Clinical Trials are commonly referred to as “post-marketing clinical trials”.
What does a Phase 3 clinical trial mean?
A study that tests the safety and how well a new treatment works compared with a standard treatment. For example, phase III clinical trials may compare which group of patients has better survival rates or fewer side effects. … Phase III clinical trials may include hundreds of people. Also called phase 3 clinical trial.
How long do Phase 2 clinical trials take?
A Phase II clinical trial lasts about 2 years. Volunteers sometimes receive different treatments. For example, a phase II trial could have 2 groups.
What is the job of a clinical researcher?
Clinical research describes many different elements of scientific investigation. It involves human participants and helps translate basic research into new treatments and information to beneficial for patients. Clinical Trials or Clinical Studies involves human volunteers.
How long does it take to be a clinical researcher?
In order to find entry-level employment as a clinical researcher, you will need at least four years of post-secondary education.
How do I become a good clinical researcher?
- Develop your understanding of best research practices. …
- Recognize how to effectively track and interpret your data. …
- Consider the patient safety and ethical concerns involved in your research efforts. …
- Familiarize yourself with regulatory guidelines.
What happens before clinical trials?
Before doing a clinical trial, investigators conduct preclinical research using human cell cultures or animal models. For example, they might test whether a new medication is toxic to a small sample of human cells in a laboratory. … Clinical trials happen in several phases during which different questions are asked.
What are the 4 phases of FDA approval?
- Step 1: Discovery and Development.
- Step 2: Preclinical Research.
- Step 3: Clinical Research.
- Step 4: FDA Drug Review.
- Step 5: FDA Post-Market Drug Safety Monitoring.
How many patients are in a phase 1 trial?
During Phase 1 studies, researchers test a new drug in normal volunteers (healthy people). In most cases, 20 to 80 healthy volunteers or people with the disease/condition participate in Phase 1.
How much money is spent on clinical trials?
The average cost of phase 1, 2, and 3 clinical trials across therapeutic areas is around $4, 13, and 20 million respectively. Pivotal (phase 3) studies for new drugs approved by the Food and Drug Administration (FDA) of the United States cost a median of $41,117 per patient.
How many patients participate in clinical trials?
Clinical trial participation rates vary from year to year. Worldwide, 105,808 people participated in research in 2015, dropping down to 25,941 volunteers in 2016.
What are top paying jobs?
- Cardiologist. National average salary: $351,827 per year.
- Anesthesiologist. National average salary: $326,296 per year.
- Orthodontist. National average salary: $264,850 per year.
- Psychiatrist. National average salary: $224,577 per year.
- Surgeon. …
- Periodontist. …
- Physician. …
- Dentist.
How many hours does a clinical researcher work?
The work of a CRA is varied and requires a lot of travel between trial sites. It is usually a 9 to 5 job but longer hours are not uncommon. Companies often pay benefits such as car allowance, additional pay etc.
Why should I do clinical research?
Clinical trials are important for discovering new treatments for diseases, as well as new ways to detect, diagnose, and reduce the chance of developing the disease. Clinical trials can show researchers what does and doesn’t work in humans that cannot be learned in the laboratory or in animals.
Is being a CRA a good job?
If you have an interest in the medical industry but don’t want to work with patients directly, a career as a clinical research associate may be ideal for you. With a strong earning potential and the ability to impact millions of lives by working on research trials, this job can be extremely fulfilling.